Ocelli (singular: ocellus) are simple eyes and are found on many different kinds of insects (such as bees, wasps, flies and dragonflies) and spiders. In adult insects, like the beautiful Jewel wasp shown below, photographed by Johan J.Ingles-Le Nobel, there are usually three dorsal ocelli located on the top of the head: two small lateral ocelli and a larger middle one called the median ocellus.

They are most pronounced on flying insects, but they have an ancient lineage and are also found on more ‘primitive’ insects, like collembolla (springtails) which can have six to eight ocelli (see below).
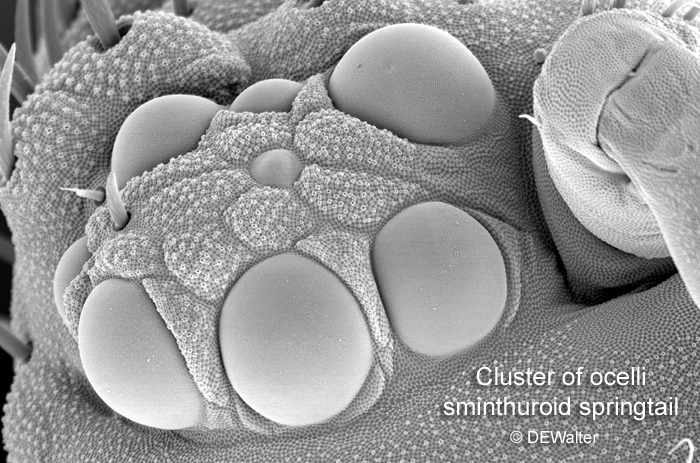
The triplet of photoreceptors on the heads of adult insects each have a lens and are used as wide-field, light detectors; but they are generally thought to be incapable of resolving images. In other words, although they have a lens which collect light, and projects it onto a layer of light-sensitive cells, the image is ‘under-focused’ and lacks detail (Krapp, 2009). Ocelli are more like a photometer – providing the insect with information on light levels – than a camera. They are very useful for telling the insect which way is up (the bright sky) and which way is down (the darker ground). Detailed resolution of images is left to the compound eyes.

In flying insects like flies, bees and dragonflies, the ocelli help the insect to ‘maintain its orientation in space; to stabilize its flight; and to keep its gaze level’ (Krapp, 2009).

Dragonflies are a bit of a special case, in that the lens of the median ocellus is ‘unusually thick and that its vertical curvature is sufficiently strong to form a focused image on the retinal receptors’ (Van Kleef et al. 2005; Strange et al., 2002).


All three ocelli are visible in the following photograph (below), with the curved median ocellus marked by the arrow.

As always, there is a lot more to this story than I have covered in this blog. Flying insects control their bodies whilst flying by evaluating a wide range of sensory inputs, including: compound eyes, ocelli, antennae, and even wind-sensitive hairs. They have had many millions of years to perfect their control systems. One advantage they get from simple eyes, is to be able to detect very fast movements, and because unlike the compound eyes, the information does not have to pass through, and be processed by the brain, they can respond very quickly: “the neurones in the median ocellus go from photoreceptor to the motor centre and they’re able to do the calculation in one step” according to Josh van Kleef.
References
Berry, R., Van Kleef, J., & Stange, G. (2007). The mapping of visual space by dragonfly lateral ocelli. Journal of Comparative Physiology A, 193(5), 495-513.
Berry, R., Stange, G., Olberg, R., & Van Kleef, J. (2006). The mapping of visual space by identified large second-order neurons in the dragonfly median ocellus. Journal of Comparative Physiology A, 192(10), 1105-1123.
Berry, R. P., Stange, G., & Warrant, E. J. (2007). Form vision in the insect dorsal ocelli: an anatomical and optical analysis of the dragonfly median ocellus. Vision research, 47(10), 1394-1409.
Krapp, H. G. (2009). Ocelli. Current Biology, 19(11), R435-R437.
Ribi, W., & Zeil, J. (2018). Diversity and common themes in the organization of ocelli in Hymenoptera, Odonata and Diptera. Journal of Comparative Physiology A, 204(5), 505-517.
Stange, G., S. Stowe, J.S. Chahl, and A. Massro. 2002. Anisotropic imaging in the dragonfly median ocellus: a matched filter for horizon detection. J. Comp. Physiol. 188:455–467.
Van Kleef, J., James, A. C., & Stange, G. (2005). A spatiotemporal white noise analysis of photoreceptor responses to UV and green light in the dragonfly median ocellus. The Journal of general physiology, 126(5), 481-497.
[…] eyes, that wrap around the side of the head, but also three simple eyes tucked in under the vertex, the ocelli. Common to many ancient insects, the ocelli have a role in keeping the insect orientated in space […]
That is incredible! thank you for sharing this.
[…] wasps, flies and dragonflies can have ocelli, although some display them more prominently than others – but look out for them next time you see some lovely insect macro photography, or get really up […]