
In this blog, I have delved into the voluminous research on the common fruit fly, or vinegar fly, Drosophila melanogaster, and tried to produce a readable, hopefully entertaining, fly-level perspective, of their courtship behaviour. I am not an expert on this species, but I have immersed myself in the literature whilst researching a book on insect courtship. I hope that I have captured the essential behaviour, without overwhelming the reader with technical terms, and that it is an interesting read! I would welcome any feedback from Drosophila researchers!

Let us start by imagining two flies – a male and a female – placed together in a small, well-lit observation chamber: a mating arena. He is naive, in the sense of never having encountered a female before, but sexually mature. She is a sexually receptive virgin, who has also had time to mature. They are both choosy, in terms of whom they mate with, but they contain all the necessary information to carry out a successful courtship, assuming they like each other! More emphasis is placed on the male’s behaviour, but only because his courtship actions are more apparent than those of the female. There are six behavioural subroutines in the courtship sequence of this species:-
- Orientation
The male Drosophila senses something moving in his visual field: photoreceptors in his eyes convey information to the optic lobe, and then on to motion-sensitive neurons in his brain, which are triggered by movements of the female; in particular her gait and pattern of activity. He approaches and stops a few millimeters away from her. Getting close to each other in this way allows both the male and the female to sense one another, particularly by receiving acoustic stimuli and detecting airborne pheromones.
The 800 facets in each of the male’s compound eyes – each one a single pixel of information – produce a fuzzy, low resolution, image of the female. Signals flash up and down nerves between his eyes and a cluster of neurons in his brain: just 20 little nerve cells in each hemisphere, which process and integrate colours, patterns and movements. The male must decide whether to initiate courtship. Other areas of the brain create a more accurate image, fly-like in size and shape. This cerebral hypothesis – together with other olfactory cues and acoustic sounds the female may be generating – triggers an attraction in the male, and he moves forward towards her. He stands facing her, separated by about 0.2 mm; she is about 2.5 mm in length, significantly larger than him.
At first, the female moves away from the male, but he persists, so she flicks out a wing, producing a sharp sound, and kicks out at him with her hind legs! She also curves her abdomen downwards to protect her genitalia from his attentions. A female who has already mated would reject the males’ advances in no certain terms by extending her ovipositor as he approached her from behind; but here, this virgin fly signals her initial rejection by kicking and repeated wing flicking. She may also ward him off by extending one or two of her legs, on the side nearest the male.
Despite these initial rejections, the male continues to follow and circle the female, tying to overcome her resistance; and she continues to move away whilst at the same time assessing him and evaluating his potential. Mating is costly for females, and she wants to make sure that coupling with this male will be sufficiently worthwhile for her. Olfactory receptors located on hair-like sensilla on her antennal lobe – and on her maxillary palps – detect odours emanating from the male. She starts to smell the volatile male pheromone which activates special neural circuits in her brain that gradually increase her receptivity and enhance her sexual motivation. Likewise, olfactory receptors on the male, and taste receptors on his foreleg, detect potent pheromone signals emanating from the female. Minute amounts of these pheromones activate courtship circuits in both of their brains.
She evaluates the male – pulling together all that she has seen, heard, smelt and tasted of him during their brief encounter – and gradually becomes more receptive, eventually stopping her movements and coming to a standstill. During the proceeding courtship contacts and posturing, she has been assessing the delicate balance of cuticular hydrocarbons, especially his principal cuticular pheromone, which she detects via her antennae, still deciding whether to not to mate with him.

2. Tapping
The male reaches out and touches the female on her abdomen with his forelegs, and then with his labial palps, bringing special taste bristles into contact with the unique blend of cuticular hydrocarbons on the surface of her cuticle. She may reciprocate, tapping him back. Chemoreceptors on the tarsi of his forelegs, enable him to determine much about her: her sex, species, age, sexual experience, and so on. The chemical compounds received from the female are also transferred to tiny taste hairs on his wings, during short bouts of grooming, which punctuate courtship behaviour. The gustatory (=taste) neurons in the contact taste receptors around his body, send all this information to his brain. It’s all written on her cuticle, so tapping her gently enables him to decide whether to continue with courtship, or to make a graceful withdrawal! In this case, the neurons in his central nervous system are activated, and trigger the ‘on’ switch for courtship to proceed, and he eagerly starts to sing!
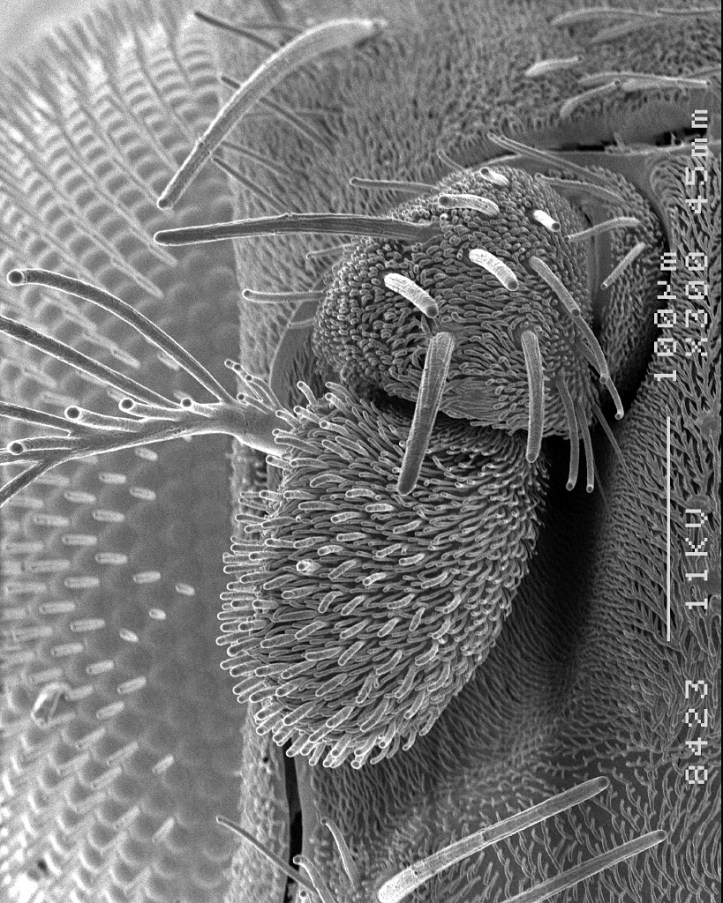
3. Singing
The male starts to sing by extending one of his wings and vibrating it. A small region at the back of his brain triggers neurons in his thorax which modulate the shape and tempo of his song pulses. Trains of loud – at least to the nearby female – but highly localised sound pulses, like a battery of rapid clicks, are occasionally interrupted by a softer humming sound: the two acoustic syllables of the song creating a species-specific chanson with a unique interval between the pulses.
The female starts to listen closely and detects a rhythmic modulation in the tiny intervals between the pulses, which fluctuate in length in a periodic manner, every minute or so. She is stimulated by the rhythmic cycles within his songs. The male continues singing, breaking off now and then after a few seconds, before resuming his acoustic vibrations, which continue in phase with the previous bout. The act of singing also has a curious effect on him; he feels turned on by his own song and enthusiastically continues courting the female!
Vibration-sensitive neurons on the female’s antennae – her love song receptors – pick up the male’s song. Her antennal ears are tuned to detect the courtship song frequency at close range. The acoustic signals race to her brain where she processes the frequency of the pulses, and the intervals between them, searching to see whether they match her built-in pattern recognition for their species.
She tunes in to the song and instinctively recognises a relatively constant interpulse interval – c. 35 microseconds for Drosophila melanogaster – filtering out frequencies that are not those of her own species. She will not mate with a fly singing the wrong song! A small set of neurons, her pulse song detectors, register the length of the pulses. She likes an energetic songster! A male whose songs have lots of long pulse trains will she thinks, be a vigorous mating partner: he who sings longer and louder should be fitter, in both an athletic and genetic sense!

For the male, his song is something he knows intuitively; it is coded in his DNA. He did not need to learn how to sing, and he can vary the patterns and amplitude of his singing in response to feedback from the female. Placing himself close to her, whilst singing his songs, is crucial for achieving a successful courtship. He circles her, sometimes singing in front, and sometimes singing behind her, positioning himself to best direct his songs towards her antennae. If she moves away from him, he starts to sing louder by including more pulse song, when she comes closer, he sings more softly. He also quivers his abdomen, generating rapid substrate-borne vibrations. The female senses these ground waves through her highly sensitive antennal ears and turns towards the source of the song (if it is in front of her) or turns away from it (if it is behind her).
Gradually, the song vibrations work their magic, changing her receptivity, and she starts to slow down, pausing every now and again, before eventually coming to halt. Eventually, as she becomes receptive, she will open her cuticular vaginal plates to allow the male access to the genitalia. For now, she listens to his song, and evaluates his pheromones, before deciding how to respond. If she doesn’t like what she sees, hears and smells, she will kick out at him once more, but in this case, she is attracted by his sensory signals, and orientates towards him.

4. Licking
The male unfurls his proboscis and ‘licks’ the female’s genitalia, bringing his labellum into contact with her ovipositor whilst placing the tips of his legs on either side of her abdomen. This upward licking motion lasts about half a second and allows him to sample a potentially different set of cuticular hydrocarbons to those located elsewhere on her body. She responds to the male’s licking by raising her abdomen.
Neurons, extending all the way down from the brain and out in branches leading to the head, thorax and abdomen, send messages to these regions triggering specific behaviours. As the gamut of sensory information is gathered and accumulated by the male, levels of excitation within his brain, steadily build up. At specific points, or sensory thresholds, various courtship elements are triggered: first proboscis extension, then abdomen curling and finally leg lifting. One activity follows the other, triggering a hierarchy of courtship behaviour.
She listens to his songs and senses his abdominal vibrations. She recognises the pattern and likes what she hears and senses. It chimes with something deep in her own DNA: recognition of one of her own kind (species). She slows down and pauses, stopping to allow the male to catch up, whilst simultaneously increasing the amount of abdominal grooming. She may still kick out at the male before becoming fully receptive, but when she does acquiesce, she spreads her wings and presents her abdomen, whilst opening her genital plates to allow intromission. Indeed, opening of the vaginal plates by the female can occur throughout courtship.
5. Attempted copulation
The male, who has been doing his best to maintain an optimal auditory signal – starts to bend his abdomen in an attempt at copulation. After licking the female’s genitalia, he attempts to grab hold of her with his forelegs, but this first attempt is usually firmly rejected by means of a vigorous backwards kick of her hind legs! Nevertheless, he persists, and the courtship sequences described above, are repeated. The male continues to curl his abdomen toward the female’s genitalia, and mating usually occurs after several such copulation attempts. Overt courtship interactions only occur for approximately 60-80% of the time; the male may cease courting for some moments, depending on the degree of his sexual enthusiasm.

6. Copulation
When and if, the female becomes fully receptive, i.e. she has responded positively to the male’s courtship stimuli, she spreads both of her wings and allows the male to mount her; both partners facing the same way as the male thrusts his abdomen towards her. The male holds the females in the correct position using special sex combs on his forelegs, and the female’s vaginal teeth interlock with the male’s claspers, keeping the pair tightly coupled. The courtship may have taken a few minutes, maybe longer, to reach this final stage and the pair will remain in copula for about 10 to 20 minutes, or so. The male transfers more than 200 different seminal fluid proteins to female, within three minutes of mating. The overall duration of courtship is very variable, depending on the motivational status of both individuals, and individual elements may be repeated numerous times before copulation takes place.


Based on the following references:
Alt, S., Ringo, J., Talyn, B., Bray, W., & Dowse, H. (1998). The period gene controls courtship song cycles in Drosophila melanogaster. Animal behaviour, 56(1), 87.
Amrein, H. (2004). Pheromone perception and behavior in Drosophila. Current opinion in neurobiology, 14(4), 435-442.
Anholt, R. R. (2020). Evolution of Epistatic Networks and the Genetic Basis of Innate Behaviors. Trends in Genetics, 36(1), 24-29.
Aranha, M. M., & Vasconcelos, M. L. (2018). Deciphering Drosophila female innate behaviors. Current opinion in neurobiology, 52, 139-148.
Batchelor, A. V. & Wilson, R. I. (2019). Sound localization behavior in Drosophila melanogaster depends on inter-antenna vibration amplitude comparisons. Journal of Experimental Biology, 222(3), jeb191213.
Baxter, C. M., Barnett, R., & Dukas, R. (2015). Effects of age and experience on male mate choosiness. Ethology, 121(4), 353-363.
Bennet-Clark, H. C., & Ewing, A. W. (1970). The love song of the fruit fly. Scientific American, 223(1), 84-93.
Billeter, J. C., & Levine, J. D. (2014). Neurogenetics: sex and the female brain. Current Biology, 24(17), R812-R814.
Billeter, J. C., & Wolfner, M. F. (2018). Chemical cues that guide female reproduction in Drosophila melanogaster. Journal of chemical ecology, 44(9), 750-769.
Bontonou, G. & Wicker-Thomas, C. (2014). Sexual communication in the Drosophila genus. Insects, 5(2), 439-458.
Branson, K., Robie, A. A., Bender, J., Perona, P., & Dickinson, M. H. (2009). High-throughput ethomics in large groups of Drosophila. Nature methods, 6(6), 451.
Bray, S. & Amrein, H. (2003). A putative Drosophila pheromone receptor expressed in male-specific taste neurons is required for efficient courtship. Neuron, 39(6), 1019-1029.
Brüggemeier, B., Porter, M. A., Vigoreaux, J. O., & Goodwin, S. F. (2018). Female Drosophila melanogaster respond to song-amplitude modulations. Biology open, 7(6), bio032003.
Bussell, J. J., Yapici, N., Zhang, S. X., Dickson, B. J., & Vosshall, L. B. (2014). Abdominal-B neurons control Drosophila virgin female receptivity. Current Biology, 24(14), 1584-1595.
Clemens, J., Coen, P., Roemschied, F.A., Pereira, T.D., Mazumder, D., Aldarondo, D.E., Pacheco, D.A. and Murthy, M. (2018a). Discovery of a new song mode in Drosophila reveals hidden structure in the sensory and neural drivers of behavior. Current Biology, 28(15), 2400-2412.
Clemens, J., Deutsch, D., Thiberge, S. Y., & Murthy, M. (2018b). Shared song object detector neurons in Drosophila male and female brains drive divergent, sex-specific behaviors. bioRxiv, 366765.
Clowney, E. J., Iguchi, S., Bussell, J. J., Scheer, E., & Ruta, V. (2015). Multimodal chemosensory circuits controlling male courtship in Drosophila. Neuron, 87(5), 1036-1049.
Cobb, M. (2019). Drosophila Courtship: Neuronal Coordination of Behavioural Sequences and a 60-Year-Old Hypothesis. Current Biology, 29(7), R250-R252.
Cobb, M., & Jallon, J. M. (1990). Pheromones, mate recognition and courtship stimulation in the Drosophila melanogaster species sub-group. Animal Behaviour, 39(6), 1058-1067.
Coen, P., & Murthy, M. (2016). Singing on the fly: sensorimotor integration and acoustic communication in Drosophila. Current opinion in neurobiology, 38, 38-45.
Connolly, K., & Cook, R. (1973). Rejection responses by female Drosophila melanogaster: their ontogeny, causality and effects upon the behaviour of the courting male. Behaviour 44(1-2), 142-165.
Dickson, B. J. (2008). Wired for sex: the neurobiology of Drosophila mating decisions. Science, 322(5903), 904-909.
Dukas, R. & Scott, A. (2015). Fruit fly courtship: The female perspective. Current Zoology, 61(6), 1008-1014.
Ejima, A. (2015). Pleiotropic actions of the male pheromone cis-vaccenyl acetate in Drosophila melanogaster. Journal of Comparative Physiology A, 201(9), 927-932.
Ejima, A., & Griffith, L. C. (2007). Measurement of courtship behavior in Drosophila melanogaster. Cold Spring Harbor Protocols, 2007(10), pdb-prot4847.
Ejima, A., & Griffith, L. C. (2008). Courtship initiation is stimulated by acoustic signals in Drosophila melanogaster. PloS one, 3(9).
Ellendersen, B. E., & von Philipsborn, A. C. (2017). Neuronal modulation of D. melanogaster sexual behaviour. Current opinion in insect science, 24, 21-28.
Ewing, A. W. (1978). The antenna of Drosophila as a ‘love song’ receptor. Physiological entomology, 3(1), 33-36.
Ewing, A. W., & Bennet-Clark, H. C. (1968). The courtship songs of Drosophila. Behaviour, 31(3-4), 288-301.
Fan, P., Manoli, D.S., Ahmed, O.M., Chen, Y., Agarwal, N., Kwong, S., Cai, A.G., Neitz, J., Renslo, A., Baker, B.S. and Shah, N.M. (2013). Genetic and neural mechanisms that inhibit Drosophila from mating with other species. Cell, 154(1), pp.89-102.
Ferveur, J. F., & Cobb, M. (2010). Behavioral and evolutionary roles of cuticular hydrocarbons in Diptera. Insect Hydrocarbons: Biology, Biochemistry and Chemical Ecology, 325-343.
Gerber, B., Stocker, R. F., Tanimura, T., & Thum, A. S. (2009). Smelling, tasting, learning: Drosophila as a study case. In Chemosensory systems in mammals, fishes, and insects (pp. 187-202). Springer, Berlin, Heidelberg.
Greenspan, R. J. (1995). Understanding the genetic construction of behavior. Scientific American, 272(4), 72-78.
Griffith, L. C. & Ejima, A. (2009a). Courtship learning in Drosophila melanogaster: diverse plasticity of a reproductive behavior. Learning & Memory, 16(12), 743-750.
Griffith, L. C. & Ejima, A. (2009b). Multimodal Sensory Integration of Courtship Stimulating Cues in Drosophila melanogaster: Contextual Effects on Chemosensory Cues. Ann N Y Acad Sci. 2009 July ; 1170: 394–398.
Grillet, M., Dartevelle, L., & Ferveur, J. F. (2006). A Drosophila male pheromone affects female sexual receptivity. Proceedings of the Royal Society B: Biological Sciences, 273(1584), 315-323.
Hall, J. C. (1994). The mating of a fly. Science 264, 1702-1714.
Han, K. A., & Kim, Y. C. (2010). Courtship behavior: the right touch stimulates the proper song. Current Biology, 20(1), R25-R28.
He, Z., Luo, Y., Shang, X., Sun, J.S. and Carlson, J.R. (2019). Chemosensory sensilla of the Drosophila wing express a candidate ionotropic pheromone receptor. PLoS Biol 17(5): e2006619.
Kallman, B. R., Kim, H., & Scott, K. (2015). Excitation and inhibition onto central courtship neurons biases Drosophila mate choice. Elife, 4, e11188.
Kimura, K. I., Sato, C., Yamamoto, K., & Yamamoto, D. (2015a). From the back or front: the courtship position is a matter of smell and sight in Drosophila melanogaster males. Journal of neurogenetics, 29(1), 18-22.
Kohl, J., Huoviala, P., & Jefferis, G. S. (2015). Pheromone processing in Drosophila. Current opinion in neurobiology, 34, 149-157.
Kyriacou, C. P. & Hall, J. C. (1980). Circadian rhythm mutations in Drosophila melanogaster affect short-term fluctuations in the male’s courtship song. Proceedings of the National Academy of Sciences, 77(11), 6729-6733.
Laturney, M, & Billeter, J. C. (2014). Neurogenetics of female reproductive behaviors in Drosophila melanogaster. In Advances in genetics (Vol. 85, pp. 1-108). Academic Press.
McKellar, C. E., Lillvis, J. L., Bath, D. E., Fitzgerald, J. E., Cannon, J. G., Simpson, J. H., & Dickson, B. J. (2019). Threshold-based ordering of sequential actions during Drosophila courtship. Current Biology, 29(3), 426-434.
Mattei, A. L., Riccio, M. L., Avila, F. W., & Wolfner, M. F. (2015). Integrated 3D view of postmating responses by the Drosophila melanogaster female reproductive tract, obtained by micro-computed tomography scanning. Proceedings of the National Academy of Sciences, 112(27), 8475-8480.
Mishra, A., & Zars, T. (2018). Sex: The End Is All You Need. Current Biology, 28(10), R614-R616.
Morley, E. L., Steinmann, T., Casas, J., & Robert, D. (2012). Directional cues in Drosophila melanogaster audition: structure of acoustic flow and inter-antennal velocity differences. Journal of Experimental Biology, 215(14), 2405-2413.
Morley, E. L., Jonsson, T., & Robert, D. (2018). Auditory sensitivity, spatial dynamics, and amplitude of courtship song in Drosophila melanogaster. The Journal of the Acoustical Society of America, 144(2), 734-739.
Paulk, A., Millard, S. S., & van Swinderen, B. (2013). Vision in Drosophila: seeing the world through a model’s eyes. Annual review of entomology, 58, 313-332.
Pavlou, H. J. & Goodwin, S. F. (2013). Courtship behavior in Drosophila melanogaster: towards a ‘courtship connectome’. Current Opinion in Neurobiology, 23(1), 76-83.
Rendel, J. M. (1945). Genetics and cytology of Drosophila subobscura. Journal of Genetics, 46(2-3), 287.
Rings, A., & Goodwin, S. F. (2019). To court or not to court–a multimodal sensory decision in Drosophila males. Current opinion in insect science 35, 48.
Ritchie, M. G., Halsey, E. J., & Gleason, J. M. (1999). Drosophila song as a species-specific mating signal and the behavioural importance of Kyriacou & Hall cycles in D. melanogaster song. Animal behaviour, 58(3), 649-657.
Sawala A. & Gould, A.P. (2017) The sex of specific neurons controls female body growth in Drosophila. PLoS Biol 15(10): e2002252.
Sethi, S., Lin, H. H., Shepherd, A. K., Volkan, P. C., Su, C. Y., & Wang, J. W. (2019). Social Context Enhances Hormonal Modulation of Pheromone Detection in Drosophila. Current Biology, 29(22), 3887-3898.
Talyn, B. C., & Dowse, H. B. (2004). The role of courtship song in sexual selection and species recognition by female Drosophila melanogaster. Animal Behaviour, 68(5), 1165-1180.
Testa, N. D., Ghosh, S. M., & Shingleton, A. W. (2013). Sex-specific weight loss mediates sexual size dimorphism in Drosophila melanogaster. PloS one, 8(3).
Thistle, R., Cameron, P., Ghorayshi, A., Dennison, L., & Scott, K. (2012). Contact chemoreceptors mediate male-male repulsion and male-female attraction during Drosophila courtship. Cell, 149(5), 1140-1151.
Toda, H., Zhao, X., & Dickson, B. J. (2012). The Drosophila female aphrodisiac pheromone activates ppk23+ sensory neurons to elicit male courtship behavior. Cell reports, 1(6), 599-607.
Tootoonian et al., 2012). Neural Representations of Courtship Song in the Drosophila Brain. Journal of Neuroscience, 32(3):787–798.
Trott, A. R., Donelson, N. C., Griffith, L. C., & Ejima, A. (2012). Song choice is modulated by female movement in Drosophila males. PloS one, 7(9).
Vaughan, A. G., Zhou, C., Manoli, D. S., & Baker, B. S. (2014). Neural pathways for the detection and discrimination of conspecific song in D. melanogaster. Current Biology, 24(10), 1039-1049.
Villella, A., & Hall, J. C. (1996). Courtship anomalies caused by doublesex mutations in Drosophila melanogaster. Genetics, 143(1), 331-344.
Villella, A., & Hall, J. C. (2008). Neurogenetics of courtship and mating in Drosophila. Advances in genetics, 62, 67-184.
von Philipsborn, A. C., Liu, T., Jai, Y. Y., Masser, C., Bidaye, S. S., & Dickson, B. J. (2011). Neuronal control of Drosophila courtship song. Neuron, 69(3), 509-522.
von Schilcher, F. (1976a). The function of pulse song and sine song in the courtship of Drosophila melanogaster. Animal Behaviour, 24(3), 622-625.
von Schilcher, F. (1976b). The role of auditory stimuli in the courtship of Drosophila melanogaster. Animal Behaviour, 24(1), 18-26.
Vosshall, L. B., & Stocker, R. F. (2007). Molecular architecture of smell and taste in Drosophila. Annu. Rev. Neurosci., 30, 505-533.
Zhang, S. X., Rogulja, D., & Crickmore, M. A. (2016). Dopaminergic circuitry underlying mating drive. Neuron, 91(1), 168-181.
Zhang, S. X., Miner, L. E., Boutros, C. L., Rogulja, D., & Crickmore, M. A. (2018b). Motivation, perception, and chance converge to make a binary decision. Neuron, 99(2), 376-388.
Zelle, K. M., Liang, X., & Ben-Shahar, Y. (2017). Pleiotropic chemoreceptors facilitate the maintenance of signal-receptor coupling in pheromonal communication. bioRxiv, 124305.
Ziegler, A. B., Berthelot-Grosjean, M. & Grosjean, Y. (2013). The smell of love in Drosophila. Frontiers in physiology, 4, 72.
As always, most interesting.Thanks!
Thanks!
Very poetic Ray, almost lyrical in places – you really made this a fascinating read – The only additional comment I have (slightly tongue in cheek) is that you could have pointed out that Tephritids are the real fruit flies and that we should all make an effort to call Drosophila melanogaster the Vinegar fly, as you did in your introduction 🙂
[…] Another great post from Ray Cannon, this time a lyrical account of the courtship behaviour of the Vinegar Fly […]
[…] As well as odours, antennae can also detect touch, gravity, wind, and sound. One of the best-studied of all antennae, is that of the fruit fly, Drosophila melanogaster. Olfactory receptors located on hair-like sensilla on the antennal lobes detect odours and pheromones, and antennal ears , yes ears, detect acoustic signals, such as the male courtship songs, as I described in another blog: A courtship duet. […]